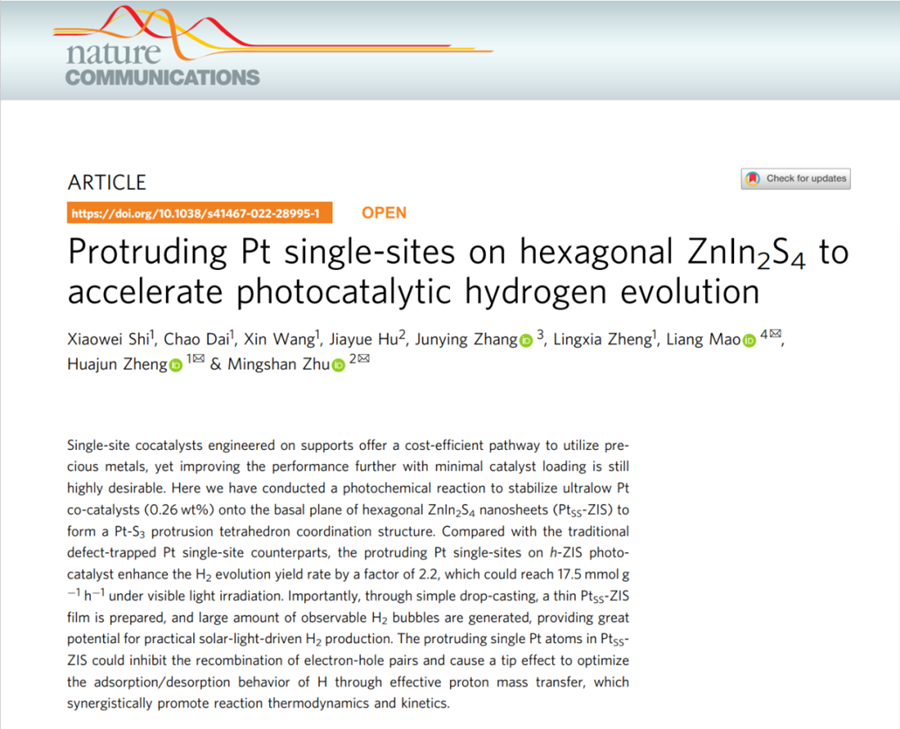
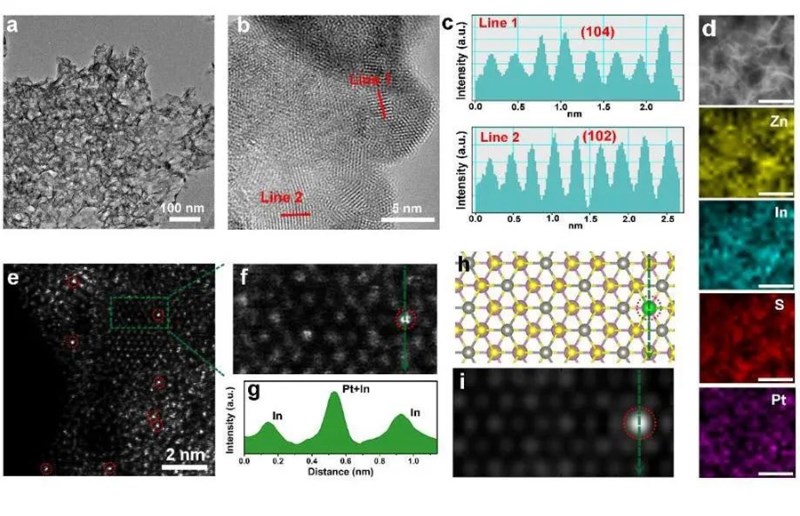
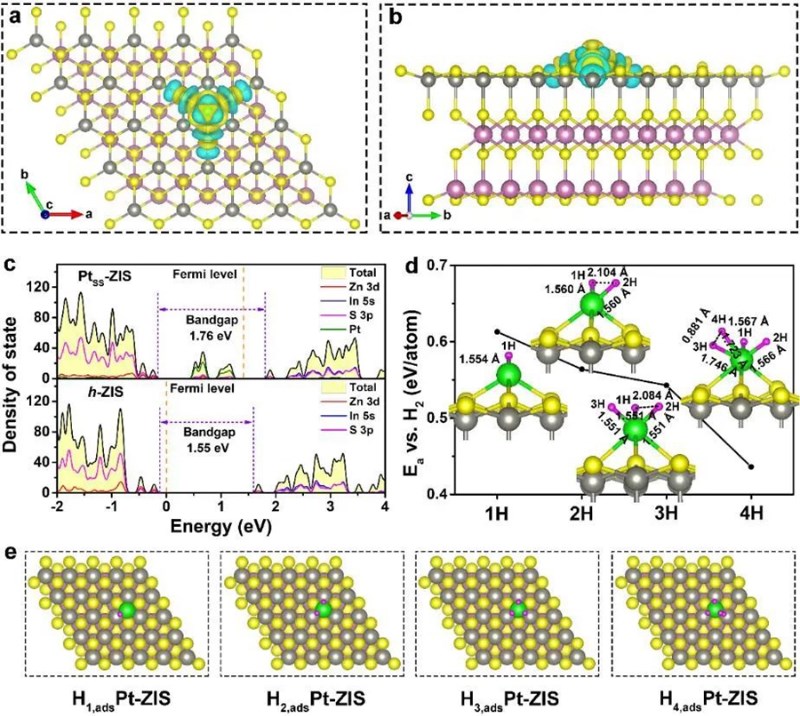
Recently, professor Zheng Huajun's team created a high-performance photocatalytic material that can split water to obtain hydrogen at room temperature. This study with the title of "Protruding Pt Single-Sites on Hexagonal ZnIn2S4 to Accelerate Photocatalytic Hydrogen Evolution" , was published online on Nature Communications on March 11, 2022.
Splitting water to generate hydrogen is the holy grail of chemists. The use of solar energy to decompose hydrogen in aquatic products at room temperature through photocatalysis is the focus of global energy scientists. High performance photocatalyst is the key to the photocatalytic cracking of aquatic hydrogen. How to use as few noble metal atoms as possible to achieve a large increase in catalytic performance is important for photocatalytic hydrogen evolution.
Professor Zheng's team has long been committed to the research of photoelectrocatalysis. The team conducted a photochemical reaction to stabilize ultralow Pt co-catalysts (0.26 wt%) onto the basal plane of ZnIn2S4 nanosheets (PtSS-ZIS). That is, in 100 mg catalyst, the mass of platinum is only 0.26 mg. Under the premise of catalyst dosage of 20 mg, the rate of hydrogen e under visible light is 350.1 millmole per hour, which is 17.8 times that of the basal plane of ZnIn2S4 nanosheets (PtSS-ZIS). Through simple drop-casting, a thin PtSS-ZIS film is prepared, and large amount of observable H2 bubbles are generated, providing great potential for practical and simulated solar-light-driven H2 production.